The Rootless Transport Mechanism
The RTM or Dr. Allen’s Procedure and the simple principal of Osmosis and Tonicity
Original article printed in Treating Yourself Magazine Issue 18, 2009 p 80
David B. Allen M.D.
Cardiothoracic and Vascular Surgeon, Member I.C.R.S. Cali215doc@gmail.com?
The Rootless Transport Mechanism or RTM procedure is a way to flavor your cannabis any flavor you choose. With this technique I make Vanilla flavored Cannabis. The RTM procedure has potential to deliver precursors of THC to the plant and allow it to make more Cannabinoids. To understand the RTM procedure you must first understand Osmosis and Tonicity.
Semi-Permeable Cell Membrane
To understand Osmosis and the concept of tonicity you have to first understand cell wall function of plants and animals. Each cell wall is composed of a phospholipid (fat) membrane which regulates the amount of fluid and salt that the cell can have in it and still have metabolic function. The cell wall has pumping mechanisms to regu- late the amount of salt content in the cell to provide opti- mal enzymatic function. Too much or too little fluid or salt will cause changes in the cell that can help or hinder cell function. The cell wall is a membrane and is called a semi-permeable membrane, because it allows some things to pass through it and keeps other things out. This means that the cell wall has the ability to pass molecules like water and other small molecules that are not highly charged and keep out large particles and highly charged particles. This semi-permeable cell membrane separates the cell from its environment and allows normal cell func-
Hypertonic Isotonic Hypotonic
tion to proceed. Without this mechanism the cell would not be able to regulate its salt and fluid content and either swell with fluid until it burst or dehydrate and shrink like a raisin. Neither of the two extremes would allow deli- cate enzymes in the cell to function. The cell actually expends a lot of its energy, by utilizing a cell wall trans- port mechanism that actively pump salts out of the cell to maintain equilibrium or homeostasis.
Tonicity
To demonstrate this phenomenon, we can see what hap- pens to a red blood cell (RBC) when placed in 3 different concentrations of salt water. When the RBC is placed in a solution of low salt
Content or pure water (this is referred to as a Hypotonic solution) the water tries to dilute out any dissolved sub- stance found inside the cell. This causes the cell to swell with the incoming water and eventually burst. (this is seen in the far right example of a Hypotonic solution.)
When the cell is in a solution where the concentration of salt is the same both inside and outside the cell, then that fluid is referred to as isotonic fluid and this fluid will not cause a net movement of water into or out of the cell. This is ideal and allows the cells metabolic machinery to function properly. (This is demonstrated in the middle example of a RBC in an isotonic solution above.)
The Third possibility is a cell in
a solution that has much more dissolved salts in the solution than what is present inside the cell (This is a Hypertonic
solution) and this causes the water in the cell to move towards a higher
con- centration of salt in the surrounding solution and causes
the cell to shrink. This is not good for the metabolic machinery to function
properly and can cause cell death
from dehydration of the cell. (This is
demonstrated in the left side of the example on the left.)
Osmosis
To understand osmosis you have to be familiar with a few terms. A solute is a solid substance that can be dissolved in a liquid called a solvent. Salt and sugars are common solutes and water is the universal solvent. The combina- tion of a dissolved salt in a solvent forms a solution. The dissolved salt is considered ionized and has a charge asso- ciated with it. Given enough time, the dissolved salt will distribute itself to a uniform concentration through-out the entire volume of the liquid. This will happen even without stirring or agitation of the solution. This is called “Even distribution of the solute in solution”
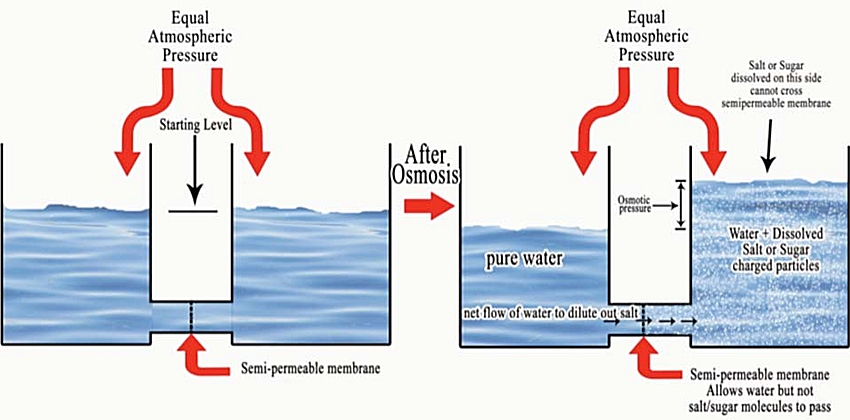
Osmosis simply stated; Is the process of a solvent (water) passing through a semi-permeable membrane from a low concentration of dissolved salt to a higher concentration of salt. In effect the water molecules try to dilute out the high concentrations of salt. The Solvent dilutes out the solute. This process continues until atmospheric pressure or other force opposes the osmotic pressure. The classic diagram of this is depicted below by two open beakers of water separated by a semi-permeable membrane. Initially the beakers are filled with water and since water can pass the membrane both containers will equilibrate to the same volume. Atmospheric pressure being the same in both beakers will cause this equilibration. If you add salt or sugar or other dissolved solute on the right beaker and allow osmosis to occur then; The solvent (Water) on the side with less dissolved salt or sugar will go to the side with a higher concentration of solute. Note the change in height of the two columns of fluid, this height difference is referred to as the osmotic pressure. If you add more salt or sugar to the right side of this example, it adds more osmotic “load” and this will increase the net water flow to the right beaker and increases the volume on that side.
This causes a net movement of water towards the higher concentration of solute, until the atmospheric pressure is equal to the osmotic pressure of the solute. (salt)
Now that we have the science down, we can have fun with the plants.
The plants roots are a semi-permeable membrane that allows water, small particles and particles with minimal electrostatic charges, to pass thru the root membrane and enter the plant. So water and other small molecules will pass into the plant and larger highly charged molecules stay in the soil. This filtering mechanism is what the roots use to protect the plant from the “shit” in the soil. (Literally and figuratively) This mechanism allows the plant to regulate the concentration of salts and water in the xylem or blood of the plant. Too many charged par- ticles in the soil act like a salt and would cause an osmot- ic pressure and in effect draws water toward it. This is exactly why putting too much fertilizer in the soil will kill the plant. The high concentration of charged particles (salt), sucks the water out of the plant and it dies of dehy- dration.
The Rootless Transport Mechanism
(RTM procedure)
I was doing and experiment trying to make root beer fla- vored pot. I thought that by putting a dilute solution of root beer in the soil that the roots would take it up. The plant was killed in my experiment, and so I had to find another way to solve this problem.
I smoked a big fatty and
re-thought my experiment to myself; I reasoned that Christmas trees once cut, can
be kept from dehydration by placing a freshly cut trunk in water and
sugar solution. I also had seen
grade school experiments where you place
a freshly cut daisy in a solu- tion of colored dye
and the plant takes up the dye and becomes the same color as the dye. The
dye is sucked up by capillary action into the leaves of the plant.
CULTIVATION
It became clear that the root of my problem was the roots of the plant. The protective mechanism that roots pro- vide, prevent any flavoring agent from entering the plant. Flavoring agents called flavanoids are large molecules that are highly charged and will not pass through the root membrane.
So I decided to try an experiment by flavoring a fully mature, ready to harvest cannabis plant with Mexican Vanilla Extract. This is an alcohol extract. I cut the mature plant trunk off just above the soil line and imme- diately placed the plant in the Vanilla Extract solution. Within 24 hours you could smell the vanilla in the bud. I was surprised as how quickly the plant sucked up the Vanilla solution and had to refill the jar several times. I left the solution for about 1 week and it took about 2 extra weeks of drying. The buds were a beautiful brown color with no green and were fragrant of vanilla. When we tried the cannabis it didn’t make you cough and had an unusual flavor that was hard to correctly name. Everyone loved it.
There seems to be a time frame where the plant must be exposed to the flavanoid and if less than about 4 days the cannabis had the smell but not the taste. Between 4-7 days seemed optimal for producing cannabis with good flavor and longer than that resulted in a harsher taste. Undoubtedly there will be an optimum concentration and length the plant has to be exposed to produce the best results. If the flavanoid is left too long the cannabis will be gummed up and impossible to dry out. I am sure these experiments are now being done as the news of this tech- nique spreads. I have already heard of success with Blueberry flavoring used on branches of the plant. Using this technique you can flavor any plant with anything you can think of! Coffee, Tea, Coke, Lemon, coconut, bubble gum the list is endless. Please no tobasco! Obviously no safety studies for the pyrolysis of these things exist but flavored tobacco products are already everywhere, and have set the standard.
This technique could also be used to flush plants fed too much nitrogen with water or isotonic solution and in effect be like placing the plant in the ICU. You could feed the live freshly cut plant an isotonic solution that would preserve cell function much like humans receive Ringers Lactate or Normal Saline IV infusions. Then you could experiment and add different “Herbs and Spices” to the isotonic synthetic xylem. You could add Antibiotics, Steroids, Bronchodilators or Opiates to the solution and the plant could have additional medical applications. Combination drug possibilities are endless. (Viagra??/ Boner Bud?)
The potential brilliance in this technique lies in the fact that you could give a plant the precursors to THC. If you could give the plant the building blocks to make THC then the plant could potentially produce Super Pot.
THC or tetrahydorcannabinol is the medicine in cannabis responsible for the majority of the medicinal effects. THC is a dimer (consisting of two compounds) of Phenols and Turpenes. The cannabis plant combines these two com- pounds and forms THC. The phenols and turpenes are bound together by a condensation bond. This bond is formed by the removal of a water molecule and is not a real strong bond like a covalent bond. (There is no shared electron) The bond is easily broken by heat and both compounds evaporate easily with a distinctive fragrant smell.
So if an isotonic solution was made with phenols and turpenes, the plant could potentially use its unique meta- bolic machinery to combine these and form THC in much higher concentrations.
MK ULTRA;
In 1953 the US government was seeing the effects of Soviet, Chinese, and North Korean use of mind control techniques on U.S. prisoners of war in Korea. In response the CIA started a program called MK ULTRA which sought to expand our knowledge on mind control, torture and brainwashing. Untrained CIA operatives conducted medical experiments on unknowing subjects using experimental drugs. Many drugs including; LSD, Amphetamines, alcohol, heroin, Morphine. MDMA, Mescaline, Psilocybin, Scopolamine, Marijuana, Sodium pentothal and ergine were used. For interesting reading please see MKULTRA and Manchurian Candidate on Wikipedia.
During these experiments they tried to make super pot by giving the plant precursors to THC, but these experi- ments failed because there was less understanding of the problems semi-permeable membranes posed to their experiments. Like me the government scientist put THC precursors in the soil and it killed the plant. I suspect they didn’t smoke a fatty and re-think their experiment. It has been previously quoted by government scientist, that you cannot give a cannabis plant precursors to THC and have it produce more THC. I am not sure that is true.
Banning scientific research on Cannabis is crazy. Busting the Oaksterdam University is an attack on Scientific Knowledge. The End of Marijuana Prohibition is Near. Science and Moral Right are on our side.